 
Calculate the amount of heat given off when 11 grams of manganese (Mn) is oxidized to Mn2O3(s) at standard state conditions. 2.24 Using the energy flow charts in Appendix 2.2 calculate the per capita (per person) consumption of energy in kWh and the average flow rate in kW in the. This reaction appears to violate this law because the entropy in 2 moles of. (3) The heat transfer of the irreversible process cannot be simply calculated without additional information, and it cannot be shown in the T-S diagram.Įntropy generation is another important concept in the second law of thermodynamics. The second law of thermodynamics states that the entropy of the universe is always increasing.  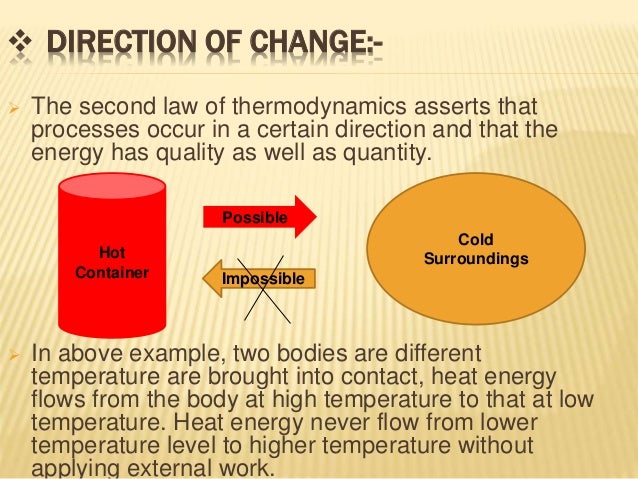
Figure 6.5.e2 T-S diagram: the shaded area represents the heat transfer of a reversible process. Record the temperature as the temperature of the air in data table 1. In principle this makes sense to me, but Im more concerned with the mathematics behind this. U is the total change in internal energy of a system, q is the. By the second law of thermodynamics, you shouldnt be able to use any amount of mirrors/lenses to focus sunlight onto an object and heat it past the surface temperature of the sun (approximately 5800K). Hold the thermometer in the air for 2 minutes, or until the reading is stable. The First Law of Thermodynamics states that energy can be converted from one form to another with the interaction of heat, work and internal energy, but it cannot be created nor destroyed, under any circumstances. The law states that it is impossible for any process to have as its sole result heat transfer from a cooler to a hotter object. This integral can be shown graphically as the shaded area under the T-S curve of the reversible process, see Figure 6.5.e2. In this experiment, we will test these theoretical predictions of Newton's Law of Cooling and the second law of thermodynamics. The Second Law of Thermodynamics (first expression): Heat transfer occurs spontaneously from higher- to lower-temperature bodies but never spontaneously in the reverse direction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
